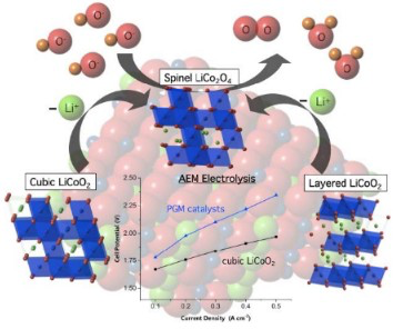
Spinel compounds like LiCo2O4 catalyzes water oxidation at lower over-potental than industry standards (Ir, Ru, Pt, Pd) . The crystal structure determines the electro-catalytic performance, with other structures (layered, cubic) or compositions (AB2O4) more useful at different current densities

Invention Summary:
Development of water oxidation/ oxygen reduction catalysts to replace costly and inefficient platinum group metals in commercial electrolyzers and fuel cells has been an unmet need preventing global development of hydrogen technologies. One limitation to electrolyzer operation is lost power efficiency due to the electrical over-potential needed to produce O2 and H2 and its strong variation with current density (flux). Both problems originate in the catalyst.
Researchers at Rutgers University have developed “designer” transition metal oxide catalysts, lithium cobalt oxide (liCo2O4) based on spinel oxides to meet these needs. Spinel oxides comprise a wide range of composition which allows tuning of the chemical, physical and electrical properties. Spinels can be tuned to achieve best electrical efficiency to match the current density needed for applications in electrolyzers and fuel cells, thereby also optimizing longevity.
This catalyst has high conductivity and is most efficient when operated at high current densities (> 400 mA/cm2) in membrane-based alkaline electrolysis. Its performance exceeds that of iridium oxide, an industry standard electro-catalyst. Comparison of spinel LiCo2O4 to the related compound LiCoO2, which can exist as both layered and cubic structure types, reveals the spinel structure type is more stable and active.
Other spinels (A ≠ Li, B ≠ Co) allow optimal performance at different electrical power, or for operation of the reverse reaction, reduction of O2 at fuel cell cathode.
Advantages:
- Lower material cost than PGM catalysts
- Lower operating cost than PGM alkaline electrodes
- Oxidation of water in the presence of the developed catalysts could lead to production of large amounts of oxygen and hydrogen
- As this process uses water as a source and sunlight as energy, it is inherently sustainable and globally scalable
Market Applications:
- Renewable O2 and H2 production Electrolyzer anode
- Counter anode for cathode reactions
- Fuel cell cathode, power generation from O2 and water
Intellectual Property & Development Status: