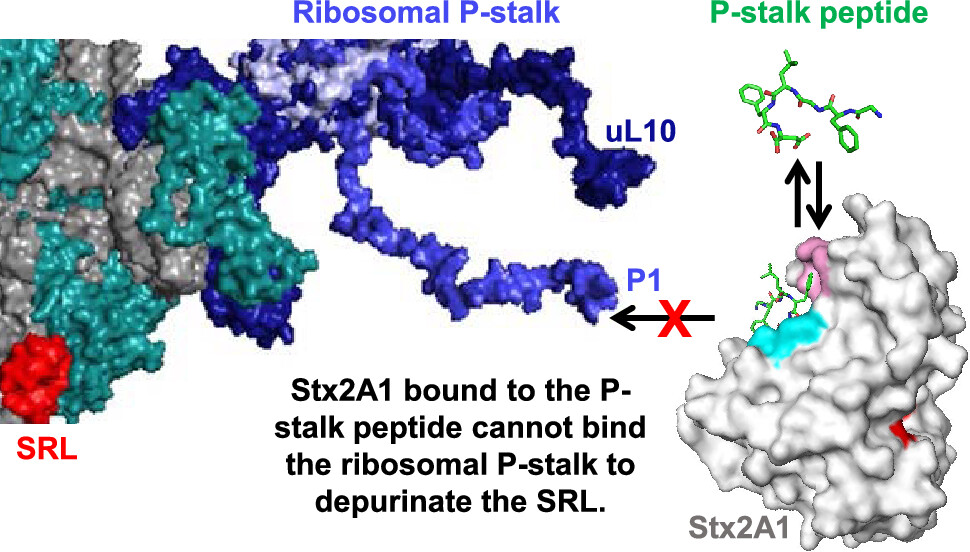

An image that shows p-stalk binding blockage would lead to the prevention of damage caused by Shiga or Ricin toxins
Invention Summary:
Shiga Toxin-producing E. coli (STEC) are very common bacteria that can cause infections that lead to approximately 100,000 illnesses, 3,000 hospitalizations, and about 90 deaths annually in the United States. Shiga toxins can cause hemorrhagic colitis and hemolytic uremic syndrome (HUS), which is the most common cause of renal failure in infants and young children in the United States. No effective therapy exists for ricin or Shiga toxin intoxication.
Rutgers researchers have identified a compound, RU-NT-206, for its high affinity in binding to the P-stalk of the ribosome, significantly inhibiting the catalytic activity of Shiga toxin 2a. This discovery stems from a fluorescence competition assay designed to screen small molecule inhibitors that can prevent the interaction between Shiga toxins and the ribosome, thereby neutralizing the toxin's harmful effects.
Market Applications:
- Pharmaceutical development for the treatment of Shiga toxin-related illnesses
- Preventative treatments in infants and young children, against HUS and renal failure
Advantages:
- High binding affinity to the P-stalk of the ribosome, approximately 60-fold higher than previous inhibitors
- Sub-micromolar potency in inhibiting the catalytic activity of Shiga toxin 2a
- Offers a potential effective therapy against Shiga toxin-induced diseases, including hemorrhagic colitis and hemolytic uremic syndrome (HUS)
Publication: Li, X.-P.; Rudolph, M. J.; Chen, Y.; Tumer, N. E. Structure–Function Analysis of the A1 Subunit of Shiga Toxin 2 with Peptides That Target the P-Stalk Binding Site and Inhibit Activity. Biochemistry 2024, 63 (7), 893-905. DOI: 10.1021/acs.biochem.3c00733.
Intellectual Property & Development Status: Provisional application filed. Patent pending. Available for licensing and/or research collaboration. For any business development and other collaborative partnerships, contact: marketingbd@research.rutgers.edu