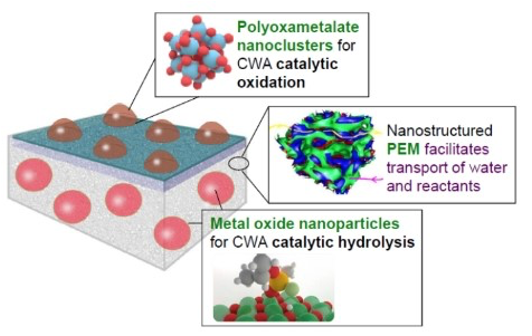
Schematic of multi-catalyst polyelectric membrane (PEM) for protection against chemical warfare agents (CWAs). Polyoxametalate nanoclusters and metal oxide nanoparticles contribute catalytic activity

Invention Summary:
To protect a wearer from toxic chemicals, such as chemical warfare agents (CWAs), materials must be both permeable to water vapor and impermeable to those CWAs. While developing such materials is a significant engineering challenge, those materials would provide only a physical barrier between the wearer and CWAs.
Researchers at Rutgers have developed a selectively permeable polyelectrolyte membrane (PEM) that includes catalytic components which decompose CWAs. The unique nanostructure of the PEM facilitates transport of water and reactants, improving the catalytic activity of the membrane. Polyoxometalates (POMs) catalyze oxidation of the CWAs, while metal oxides (MOs) catalyze hydrolysis of CWAs. This multi-catalyst polyelectrolyte membrane could be incorporated into textiles to generate self-decontaminating, protective clothing for personnel who may be exposed to harmful chemicals, including CWAs. There may be additional applications related to biomedicine and energy.
Advantages:
- Physical barrier: selective permeability
- Permeable to water vapor
- Impermeable to chemical warfare agents
- Chemical barrier: multiple catalytic components
- Hydrolysis
- Oxidation
- Transport of water and reactants
- Compliant material
Market Applications:
- Self-decontaminating, protective clothing
- Chemical warfare agents Workplace hazards
Intellectual Property & Development Status: US Patent 62/162,402; 15/154,219. Available for licensing and/or search collaboration.