Invention Summary:
Neuropathic pain (NP) typically arises as a consequence of a lesion or disease affecting peripheral nerve fibers. Prominent features in the pathophysiology of neuropathic pain include increased, ectopic neuronal activity in pain pathways. In general, NP is frequently refractory to conventional analgesics and currently there are no effective treatments.
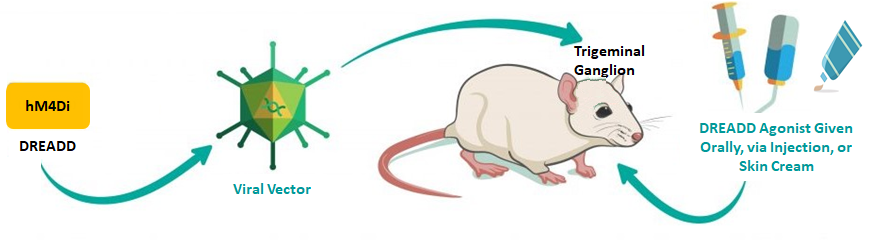
Rutgers researchers have proposed a chemogenetic therapy for NP, where a novel viral vector encoding the synthetic Gi-coupled DREADD (Designer Receptors Exclusively Activated by Designer Drugs) receptor known as hM4Di, is microinjected into the trigeminal ganglion of rat. A robust expression of the hM4Di receptor in ganglion neurons is achieved under control of the Tac1 promoter. After the hM4Di receptor is activated by the DREADD agonist, e.g. Olanzapine (marketed as Zyprexia), the indices of pain in the rat model of trigeminal neuralgia are significantly decreased.
In addition, the hM4Di receptors are transported distally along trigeminal ganglion axons, therefore, these receptors can be stimulated peripherally to achieve pain relief, e.g., by topical application of a skin cream containing the DREADD agonist. This targeted gene therapy should also be effective for other neuralgias by expressing the hM4Di receptor in the dorsal root ganglion associated with any peripheral nerve.
Advantages:
- Novel targeted gene therapy to express the hM4Di receptor in peripheral ganglion neurons and decrease their activity by administration of the DREADD agonist
- Tac1 promoter-driven vector to express hM4Di receptors specially in neurokinin neurons to limit effect of the DREADD agonist to neurons that mediate pain information
- A topical application of the DREADD agonist in a skin cream
Market Applications:
- A therapy to treat any painful peripheral never neuralgia, including trigeminal neuralgia
Intellectual Property & Development Status: Patent pending and patent publication: US 2019/0046662 A1. Available for licensing and/or research collaboration.