|
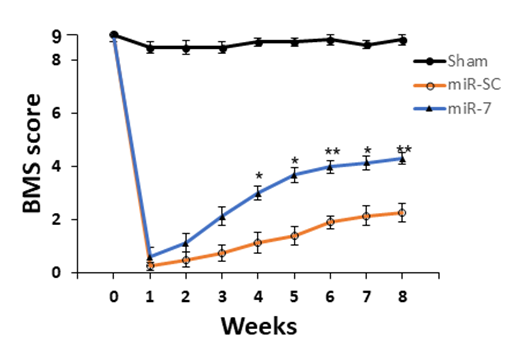
Following sever spinal cord compression, the AAV-miR-7 injected mice showed improved locomotor recovery, which is characterized by the Basso Mouse Scale (BMS).
|
Invention Summary:
Spinal cord injury (SCI) results in partial or complete loss of sensory or motor function. According to the National Spinal Cord Injury Statistical Center, more than 290,000 people are suffering from SCI in the U.S., with 17,810 new SCI cases occurring each year. Depending on the severity and level of the injury along the spine, SCI can be life-threatening. The recovery process of SCI is very slow and usually incomplete. Currently, there is no cure for SCI. Thus, effective, safe and feasible treatments are urgently needed to improve SCI recovery.
Rutgers scientists have discovered that microRNA-7 (miR-7) can improve motor functional recovery after spinal cord injury which can be exploited as gene therapy to treat SCI using an AAV vector. The inventors have shown in a mouse model of SCI that following severe compression of the cord, AAV-miR-7 transduced mice exhibit improved locomotor recovery compared to control mice with SCI that received the AAV vector without miR-7. AAV-miR-7 transduction also targeted other neuroprotective genes and signaling pathways, including reduced glial responses, increased neuronal survival, axon regeneration, and protection of oligodendroglia. Taken together, miR-7 gene therapy attenuates SCI-induced neuronal death and improves functional recovery. The use of AAV-miR-7 provides a novel therapeutic for effective early and sustained recovery from the devastating consequences of SCI.
Advantages:
- Highly effective
- Multiple neuroprotective targets
- Potential to combine with other existing treatments
Market Applications:
- Gene therapy for spinal cord injury
Intellectual Property & Development Status: Patent pending. Available for licensing and/or research collaboration.