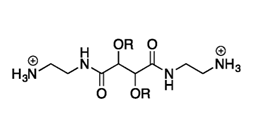
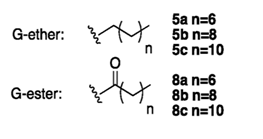
The structure of CAms 5a, 5b, 5c, 8a, 8b, and 8c
Invention Summary:
The development of antibiotic-resistant bacteria is a prevalent concern that has prompted the development of new antimicrobial agents. Naturally-occurring antimicrobial peptides (AMPs) offer advantages over conventional antibiotics due to a reduced development of resistance mechanisms through disrupting bacterial membranes. However, the therapeutic use of naturally occurring AMPs is hampered by the high manufacturing costs, poor pharmacokinetic properties, and low bacteriological efficacy in animal models.
Rutgers researchers have synthesized novel cationic amphiphiles (CAms), which mimic naturally-occurring AMPs and can be used in antimicrobial applications. The structure of CAms can be modified and their biocidal potential can be improved to counteract resistance to antibiotics. The CAms can self assemble into micelles, complex with liposomes, or be formulated into nanoparticles for various delivery applications. These CAms could thus be used as therapeutic compounds, as delivery vehicles for bioactive, including oligonucleotides, or for diagnostics.
Overall, these CAms could improve the performance of current antimicrobial products or be competitive with existing antimicrobial technologies.
Advantages:
- Have the potential of mitigating drug-resistant bacterial and may not lead to development of resistance
- Easier and cheaper to synthesize relative to peptides
Market Applications:
- Potential new class of antibiotics for therapeutic use
- Novel antibiotic materials for personal care products (soaps and detergents)
Intellectual Property & Development Status: US patent: 10,759,740. Available for licensing and/or research collaboration.
Publication: